By Levi Welch, Harris Westminster Sixth Form
Citation
Welch, L. (2022) The Great Oxidation Event: A case study of the relationships between geographical and biological systems. Routes 3(1): 4-11
Abstract
This essay aims to highlight the interlinked nature of geographical and biological systems and the great complexity of change these systems have over time. It will follow the story of cyanobacteria and how their evolution has had consequences for geology to biodiversity. This includes the formation of iron banded rock due to reactions between iron in the ocean, and cyanobacteria’s waste product, oxygen. This essay will also consider the effect that oxygen had on lowering temperatures to allow a global glaciation, and the opportunity unlocked by oxygen’s presence for the evolution of complex life.
Key words
Cyanobacteria – A group of related bacterial microorganisms capable of photosynthesis.
Phylogenetic – Relating to the evolutionary history of a group of organisms and the evolutionary relationships between organisms.
Proterozoic – A geologic eon spanning 2.5 billion years ago to 0.54 billion years ago.
Glaciation – The process of an area of land or ocean being covered by glaciers or ice sheets.
Anaerobic – Related to or requiring the absence of oxygen.
Adenosine Triphosphate – A biological molecule consisting of an adenosine bonded to three phosphates. Functions as energy storage and is used in a cells basic metabolism.
1. Introduction
2.4 Billion years ago, at the beginning of the Proterozoic Eon, the earth’s surface was covered by a vast ocean. With an atmosphere almost fully composed of nitrogen and carbon dioxide, all oxygen was locked away in the earth’s great water store.
Life at this time had only just begun to complexify and one organism was about to discover a new ecological route to success. Through a chemical alteration, it would permanently change the earth’s geology, geography, and biodiversity. This highlights the significance of the interrelationships and feedback mechanisms in both the geographical and biological systems of earth. This is clear as the vast changes that will be explored were all set in motion by a single group of organisms. The organisms in question are the microscopic cyanobacteria (see Figure 1).
2. An Increase in Oxygen
Cyanobacteria had developed a chemical niche in which it could absorb sunlight with an internal pigment, exciting electrons, which travel down transport chains, converting energy from the surroundings into useful chemicals. These chemicals would be synthesised from carbon dioxide, while water would be broken down to replace the excited electrons. This reaction required only water, carbon dioxide, and light, all three present in abundance, and so this biological invention catapulted cyanobacteria to success, its population growing subsequently. Despite the effectiveness of this new kind of energy production, there was one significant byproduct. As water was split it produced an electron (very useful), a proton (very useful), and an oxygen molecule (not so useful). Any cell engaged in metabolism must solve the problem of how to rid itself of its waste products, and so at the same time as these organisms learnt to live off the light, they also learnt to release their oxygen leftovers.
How do we know cyanobacteria were the first organisms to produce oxygen? One important piece of evidence is phylogenetic data (comparisons of the gene sequences of different bacteria). What we see is that cyanobacteria is one of the most diverse groups genetically, which suggests that cyanobacteria have had a longer period to diverge from each other, (change gene sequences by random mutation). They are therefore a very ancient group, and probably the most ancient photosynthetic organisms. If this is correct then they must be the original oxygenators. In addition to this, some very early fossils have been uncovered that seem to resemble cyanobacteria, however, this evidence can be hard to interpret and its reliability is less strong.
Initially, as oxygen was released into the surrounding ocean by cyanobacteria, very little atmospheric change resulted.
Due to the chemical composition of the ocean in the early Proterozoic, oxygen could not travel far before it was captured by some reactive molecule (Lyons et al., 2014). These molecules were available in such great abundance, as previously they had no co-reactant, and so floated passively for millions of years. One such reactant was iron, which would have acted as a major oxygen sink, binding to, and so controlling the concentration of free oxygen. These reactions formed new compounds like Hematite (Fe2O3) and Magnetite (Fe3O4), which, unlike iron, cannot dissolve in water and are much denser, so sink to the ocean bed. Over a great length of time, this reacted matter accumulates, compacts, and forms alternating layers of iron oxides (see Figure 2), that can extend several hundred metres in depth (Kappler et al., 2005).
Of course, this occurs at a rate of only 1 metre every 4,000 years. But we may view this as remarkable speed. Consider the microscopic organisms that have authored this change; 30,000 cyanobacteria would have to be placed one on top of the other to bridge the same 1 metre gap. It is quite amazing that such small creatures have made such grand formations.
The scale of oxygen sinks, such as iron, were so great that they were only depleted after 50 million years. It was only after this time that oxygen began trickling into the earth’s atmosphere.

3. Global Glaciation
Between 2.4 and 2.1 billion years ago, it is believed that the earth was covered in a sheet of ice (Marshall, 2010). Imagine our poles, the Arctic and Antarctica, spreading out from the north and south, to cover much of the globe, a snowball earth. This event coincides with the first release of oxygen so smoothly that it suggests a causative relationship.
One possibility as oxygen first filtered out of the ocean, was a large-scale reaction with methane. Methane is an extremely potent greenhouse gas, with a global warming potential 25 times higher than carbon dioxide in 100 years. It was therefore responsible for the warm conditions that allowed surface water before the Proterozoic Eon.
As oxygen began to accumulate, it may have bonded with methane, giving off the products carbon dioxide, and water. As carbon dioxide is much less potent than methane, less reflected solar radiation would be reabsorbed by the atmosphere, and so if this reaction was common enough, over a long period of time, we would see a drop in global temperatures; a fitting cause for glaciation (Mohajan, 2011).
This hypothesis highlights the importance of cyanobacteria, however, there may be other significant events that also played a role. It seems likely that astronomical changes were a factor in the temperature alterations at this time. This could have occurred through a decrease in solar output, or through changes in the eccentricity of orbit, exposing the earth to a lower intensity of sunlight. Another possibility is a decrease in the volcanic outgassing rate, leading to a lower concentration of greenhouse gases in the atmosphere (Teitler et al., 2014). This would mean that less reflected solar radiation would be reabsorbed by the atmosphere and so the earth would cool. I would suggest that an event as large as a global glaciation must have occurred as the cumulative effect of several factors, and among these factors, oxygen-methane reactions, facilitated by cyanobacteria, would have been very significant.
One problem that quickly emerges with a global glaciation can be expressed in the question “We know how to get into the ice age, but how do we get out of it?” It happens that cooling down is much easier than heating back up to pre-glaciation temperatures. Here we see the worrying influence of feedback loops.
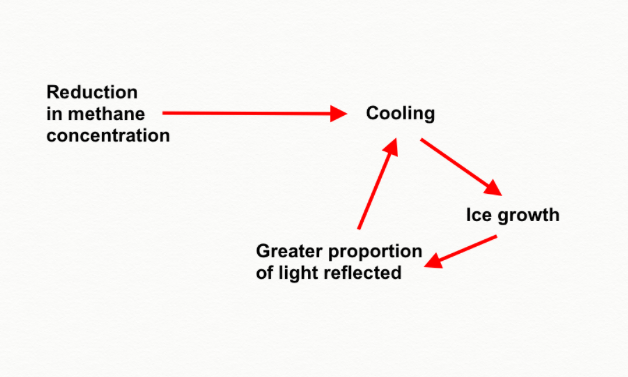
As ice on the poles extend, a greater proportion of the earth’s surface is covered in bright, white ice. This ice has a much higher albedo, (proportion of solar radiation that is reflected), than the surrounding water, and so radiation that was previously absorbed by the dark blue ocean is now being reflected away. The same process is occurring in modern time but in reverse with ice caps melting to reveal dark water, which then absorbs a larger amount of solar radiation, and so heats up more, facilitating further ice melt (Curry et al., 1995). During a global glaciation ice’s low albedo decreases the energy input to the earth and results in accelerated cooling (see Figure 3). It now becomes difficult to see how a planet that is reflecting most of the sun’s energy away, will ever heat up enough to break the surface deadlock.
One way to escape this trap could be to invoke some other powerful geological process from this period. A good candidate maybe, once again, volcanic activity. This would work through a regular spewing out of greenhouse gases over a long period of time, eventually rising temperatures enough to melt the earth’s icy surface and reveal the blue beneath (Cole-Dai, 2010).
4. A Golden Age of Biodiversity
The area in which the great oxidation event has had the most profound and far-reaching impact is the change it caused, and the evolutionary potential it unlocked, in the biodiversity of the planet’s ecosystems.
In the immediate aftermath of the great oxidation event was the earth’s first large extinction event (Ashraf et al., 2009). The atmosphere had never been so full of oxygen, and the bacteria of the ocean were not adapted to such an environment. Very quickly, huge portions of the tree of life were wiped out and today only a few anaerobic life forms remain, usually confined to areas with very low oxygen concentration like peat bogs (Duddleston et al., 2009). At the same time, a population boom in species that could convert the now abundant oxygen into energy left the ocean inhabited by new oxygen-absorbing organisms. Here we see the birth of oxygenic respiration.
One pioneer in this field may have been a bacteria we now call mitochondria. In an early form, it would have specialised in synthesising the molecule Adenosine Triphosphate (see Figure 4). ATP has the special property of being capable of powering almost all chemical processes within cells by shooting off one of its three phosphates. And so respiration handed to mitochondria a huge biological advantage.
Around 2 billion years ago, a larger cell (probably an archaeon) engulfed a mitochondrion, and by some strange, probably random adaption did not destroy it but let it go on living inside itself (Kutschera and Niklas, 2005). This was the start of probably the most successful cooperation in evolutionary history. Every single cell in your body, and every plant and animal cell, contains hundreds to thousands of mitochondria, fuelling all metabolic processes through ATP synthesis.

If it was not for the cyanobacteria, then oxygen concentration would never have increased, mitochondria then could have never evolved, and all the complexities of plants and animals would be lost with it. The great biodiversity of our ecosystems has cyanobacteria to thank. Here we see again that the consequences of cyanobacteria reach far beyond their ancient history in the oceans.
5. Conclusion
I have intended this essay to, above all, illustrate the complexity of earth’s geographic and biological systems, and the intricate ways in which they interact. We must understand the geographical nature of our world not just to investigate the past, but also to consider how the world’s oceans, atmosphere and ecosystems may change in the future. I have followed the most accepted theories surrounding the great oxidation event but we should be aware that as new evidence emerges scientific consensus shifts and so the current orthodoxy must be challenged. That is one of the jobs of the next generation of geographers, to see whether what we currently think stands to scrutiny. What is certain is that there is still so much of earth’s history to be uncovered and so much geographical complexity still to be explored.
6. References
Ashraf, M.T., Elewa, B. and Joseph, R. (2009) ‘The History, Origins, and Causes of Mass Extinctions’, Journal of Cosmology, 2, p201-220.
Cole-Dai, J. (2010) ‘Volcanoes and Climate’, WIRES Climate Change, 1(6).
Curry, J.A., Schramm, J.L. and Erbert, E.E. (1995) ‘Sea Ice-Albedo Climate Feedback Mechanism’, Journal of Climate, 8(2), p240-247.
Duddleston, K.N., Kinney, M.A., Kiene, R.P. and Hines, M.E. (2002) ‘Anaerobic microbial biogeochemistry in a northern bog: Acetate as a dominant metabolic end product’, Global Biogeochemical Cycles, 16(4), p1-11.
Kappler, A., Pasquero, C., Konhauser, K.O. and Newman, D.K. (2005) ‘Deposition of banded iron formations by anoxygenic phototrophic Fe(II)-oxidizing bacteria’, Geology, 33(11), p865-868.
Kutschera, U. and Niklas, K.J. (2005) ‘Endosymbiosis, cell evolution, and speciation’, Theory in Biosciences, 124, p1-24.
Lyons, T.W., Reinhard, C.T. and Planavsky, N.J. (2014) ‘The rise of oxygen in Earth’s early ocean and atmosphere’, Nature, 506, p307–315
Marshall, M. (2010) The history of ice on Earth [Online]. Available at: https://www.newscientist.com/article/dn18949-the-history-of-ice-on-earth/. (Accessed: 8 October 2021)
Mohajan, H.K. (2011) ‘Dangerous effects of methane gas in atmosphere’, International Journal of Economic and Political Integration, 1(2), p3-10.
Teitler, Y., Hir, G., Fluteau, F., Philippot, P., and Donnadieu, Y. (2014) ‘Investigating the Paleoproterozoic glaciations with 3-D climate modeling’, Earth and Planetary Science Letters, 395, p71-80.
#Write for Routes
Are you 6th form or undergraduate geographer?
Do you have work that you are proud of and want to share?
Submit your work to our expert team of peer reviewers who will help you take it to the next level.
Related articles
